Chemistry Cartridge CGI Examples
Daylight 4.61, dayblob 46107, Oracle Server 8.13
Chemistry Cartridge demo examples
A number of CGIs are available for demonstration of the
Chemistry Cartridge prototype.
These demos operate on the 118,616 structures in the
NCI95
database as served by a beta Oracle 8i server
and processed by the prototype chemistry cartridge v46107.
Hyperlinks on this page do not actually run the searches,
but are linked to copies of the HTML pages (including honest
timings) that resulted when run on a Sun Ultra-60 workstation
using a single 360 MHz processor.
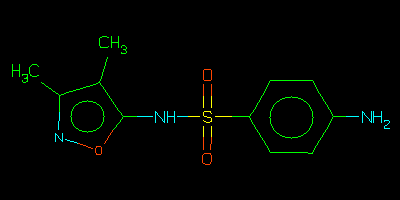
Sulfisoxazole
The sulfisoxazole structure contains many characteristic features
and is an example of a query which is particularly easy to process.
It also represents a kind of chemical information query which is
common in pharma/agro industries.
Although there are 4 superstructures of sulfisoxazole in NCI95,
there is nothing terribly similar to it (in this case, the Nearest
search is probably more useful than Similar).
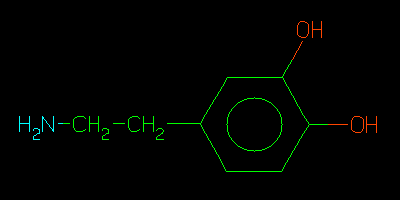
Dopamine
The dopamine structural pattern is very common in both natural and synthetic
substances. For such patterns, transferring results to Oracle server can
become rate-limiting, e.g., Contains and Match take about 2.5 seconds to
complete. This bottleneck may be removed in future versions of the Oracle
server which are expected to implement shared memory objects.
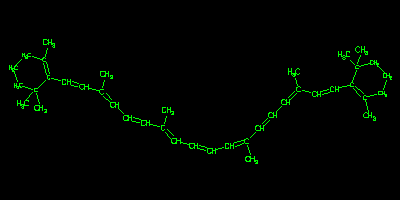
Beta Carotene
Compared to typical "small molecules", beta carotene is a large,
featureless structure. Such structures are often difficult to
process. The Daylight fingerprint methodology is extremely
efficient in extracting search features, and this shows up as
the excellent (subsecond) Chemistry Cartridge performance below.
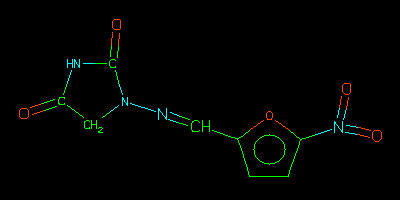
Nitrofurantoin
Tautomer lookup is typically used to find structures allowing for migration
of charges and hydrogens. In this example, an uncharged representation of
the nitrofurantoin structure was entered as a query to each search.
Nitrofurantoin is in NCI95, but in a different representation
(a charge-separated structure). Tautomer, Similar, and Nearest searches
are robust in this manner, and find it anyway.
Intra-molecular hydrogen bonds
The SMARTS O=[N,C]aa[N,O;!H0] describes a pattern of two
substituents on an aromatic ring which can form an intramolecular
hydrogen bond, something which has a profound effect on molecular
polarity and hydrophobicity. Of the database functions available
in the Chemistry Cartridge, only Matches() can be applied since
this is a SMARTS pattern. This pattern is "pathologically"
difficult to search because the only fixed part upon which the
screens can operate is O=*, a very common substructure.
Note that the time it takes to process this is very long (more
than a minute) compared to most other searches (less than a second)
Database
operation
| Number of rows
selected
| Elapsed time
min:sec
| Links
|
|---|
| MATCHES
| 4545
| 01:16:40
| SQL,
Results
|
 Daylight Chemical Information Systems, Inc.
Daylight Chemical Information Systems, Inc.
info@daylight.com


