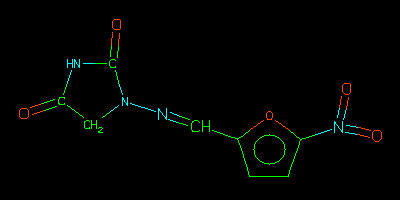
O=C1CN(N=Cc2ccc(o2)N(=O)=O)C(=O)N1
The Chemistry Cartridge adds the SIMILAR operator to SQL. When applied to a SMILES-containing column, rows containing structures which are similar to a given structure are selected. The binary Tanimoto coefficient is used for similarity determination: a value of 1.0 means "identical", 0.0 means "completely dissimliar". The level of similarity to be required is user-adjustable. At high levels (~0.8 or more), this is a fast, robust search type. At low levels (~0.65 or less), the results are not meaningful and result in large answer sets.
This example finds structures at least 0.80 similar to a given structure using svrmgr (rather than sqlplus to show the auxilliary operator score in action. Other examples are also available.

Oracle Server Manager Release 3.1.3.0.0 - Beta
(c) Copyright 1997, Oracle Corporation. All Rights Reserved.
Oracle8 Enterprise Edition Release 8.1.3.0.0 - Beta
With the Partitioning and Objects options
PL/SQL Release 8.1.3.0.0 - Beta
SVRMGR> Connected.
Timing ON
COMPOUND_I SCORE(1) SMILES
---------- ----------- ---------------------------------------
43894 0.873831749 O=C1CN(N=Cc2ccco2)C(=O)N1
55940 0.876652002 [O-][N+](=O)c1ccc(C=NN2CCNC2=O)o1
78432 0.929515421 [O-][N+](=O)c1ccc(C=NN2CC(=O)NC2=O)o1
95811 0.800000012 OCCN1CCN(N=Cc2ccc(o2)[N+](=O)[O-])C1=O
4 rows selected.
Parse 0.00 (Elapsed) 0.00 (CPU)
Execute/Fetch 0.92 (Elapsed) 0.00 (CPU)
Total 0.92 0.00
Timing OFF
SVRMGR>
Server Manager complete.