3D 3 Point Descriptors
The 3D 3 Point Descriptors are build from the 3 dimensional structure of the
compounds each feature (pharmacophore) consists of 3 atoms with associated
interaction eg. hydrogen bond acceptor, donor, hydrophobic, (pharmacophore point)
and the distances between this features. Have a look at an example:
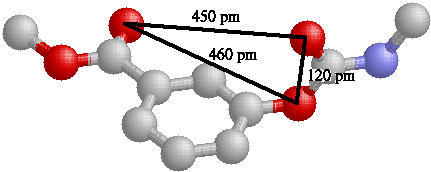
3D Pharmacophores pose quite a few challenges we have to deal with:
- The distances are continuous, this is handle by bining the distances like
bin one might be distances between 0 and 180 pm, bin two from 180 to 250 and
so on.
- A compound with routable bonds may take many different conformations and
even worst may adopt itself to fit into a receptor. To find pharmacophores
a molecule can have we need to do a conformational analysis and look at
sometimes many thousands of conformations.
- The completeness of a conformational analysis is difficult to archive.
Depending on the completeness of the conformational analysis and the number
of bins, which are used for the distances a compound will have many thousands
of pharmacophores out of a list of a few millions. Some people are now using
4 and 5 point pharmacophores which allow to distinguish chiral elements but also
greatly increase the number of pharmacophores.
Besides this problems 3D pharmacophores might have some advantages as
compared to 2D descriptors, although the literature is controvers in real
experimental results:
- The distances are real distances in 3D space, so two completely different
compounds may share the same pharmacophore.
Currently WATER does not use 3D descriptors but the implementation does not
pose any significant problem since conformational analysis algorithms are
available on the marked from various sources.
Page Owner:
Alberto Gobbi
- Last updated: Apr 17, 1999

